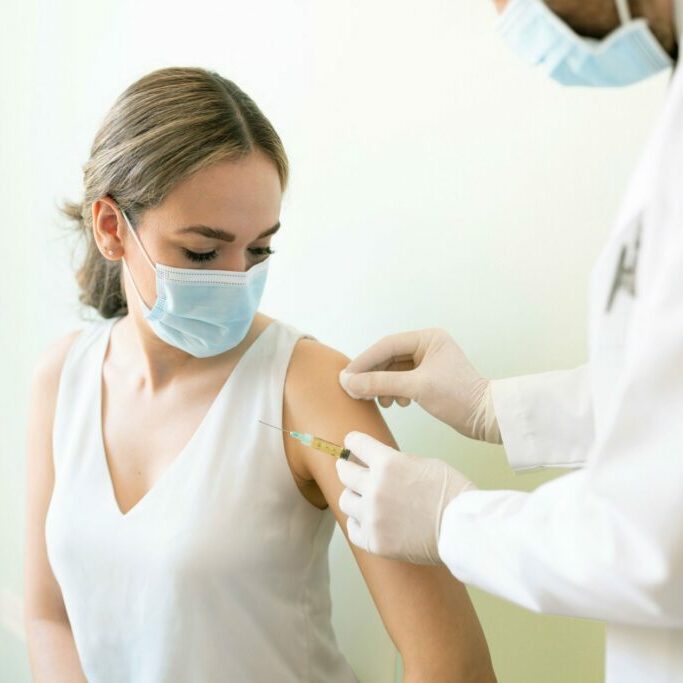
Velocity’s San Diego research site was founded in 1999 as eStudySite, by Dr. William O’Riordan, and acquired by Velocity in 2020. Through comprehensive research services agreements with each of its partner hospitals, the clinic has been able to conduct a broad range of in-patient and out-patient protocols spanning first-in-human/first-in-patient through to pivotal trials and post-marketing support. Our San Diego site has a worldwide reputation for excellence in conducting infectious disease trials including antibiotics, antivirals, vaccines, and diagnostic studies, consistently enrolling among the top 5% of participating sites. Over the last 20 years, the site has continued to grow its therapeutic area expertise to include: endocrinology, hepatology, dermatology, and gastroenterology, and has forged relationships in the community enhance recruitment of elderly and other sub-populations. Our bi-lingual research team successfully recruits and enrolls many Latinx study participants.
Accessible to several major highways, this ADA-compliant facility serves the greater San Diego area and includes an inpatient/outpatient infusion center. With more than 4,400 square-feet of dedicated research space the research team can efficiently and effectively conduct multiple concurrent studies. All trials conducted at the site are performed in accordance with ICH and FDA guidelines, and in compliance with GCP. All Nurses are ACLS certified and all other staff are BLS certified. The San Diego team is committed to being a resource for study participants, to providing the highest quality of patient care with compassion and kindness, and to advancing medicine through research.