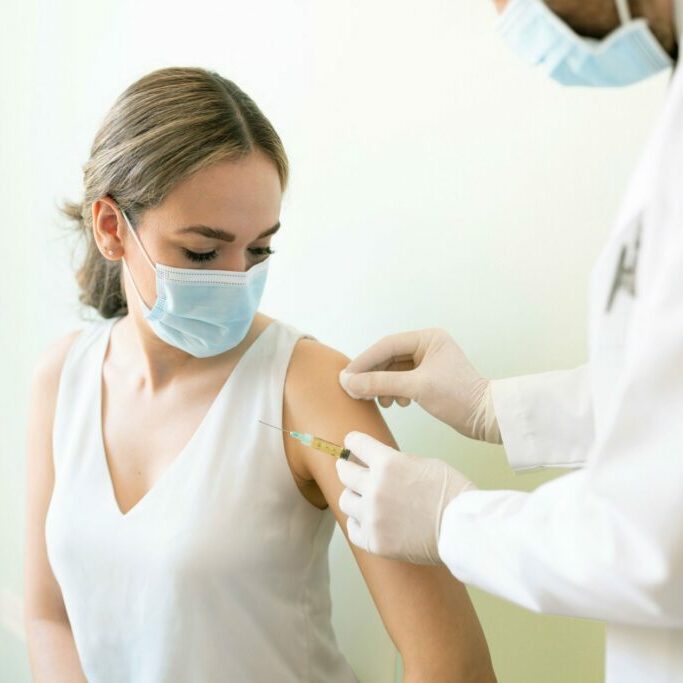
Velocity’s research site in Austin was founded as in 2019 as part of Advanced Clinical Research, and acquired by Velocity later that year. The site was started with a core team of experienced research staff from our Idaho location, and has expanded to form a robust research team. The site in Austin provides expertise in a wide range of therapeutic indications specific to the geriatric population, including dementia, osteoporosis, and other chronic conditions. By developing close working relationships with area retirement communities and having recruiters trained in the nuances of elderly participants, Austin enrolls a high number of patients over the age of 65.
Conveniently located in Austin, this site offers ease of access from major highways without the congestion of downtown traffic, as well as ample patient parking. The site is located less than one mile from EMT services and less than 5 miles from a regional hospital.
All trials are performed in accordance with ICH and FDA guidelines, and in compliance with GCP. The Austin team is committed to being a resource for study volunteers, to providing the highest quality of patient care with compassion and kindness and to advancing medicine through research.