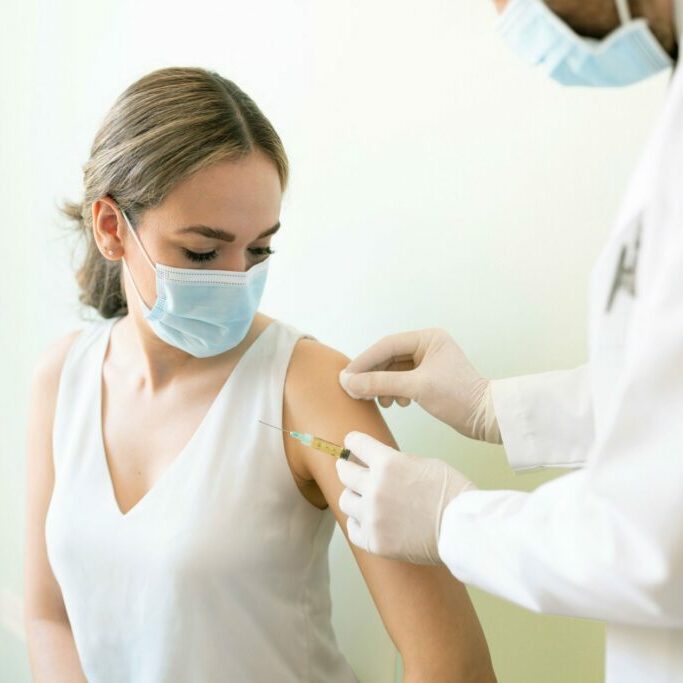
Velocity’s research site in Anderson was founded in 2013 as part of VitaLink Research, and acquired by Velocity in 2021. The Anderson location has a strong track record of success with respiratory trials, including COPD and Asthma. In recent years the site has worked to become more therapeutically diverse, with trials in diabetes, Alzheimer’s, and a variety of vaccines. All trials are performed in accordance with ICH and FDA guidelines, and in compliance with GCP.
Conveniently located near Downtown Anderson, this site shares a building with a Pulmonology office providing the research team with easy access to potential patients. Ample patient parking is also available. Our Anderson site is located just over one mile from the local hospital. The Anderson team is committed to being a resource for study volunteers, to providing the highest quality of patient care with compassion and kindness and to advancing medicine through research.